Belviq Review (UPDATED 2024): Don't Buy Before You Read This!
The prescription weight loss drug officially named Lorcaserin that is FDA approved.
Top Rated Diet Pills of 2024What is it?
Belviq is a weight loss diet pill sold via prescription that’s made to help suppress appetite. It produces this affect by changing receptors in the brain, thereby reducing cravings for food.
It’s only advised for use among healthy people without certain conditions, and alongside a diet and exercise plan. Only people with a BMI of 30 or more, or with a BMI of 27 or over if one suffers from an obesity related illness can take Belviq. Clinical studies have been performed for over 2 years, with some showing a 5% reduction in weight in the first year. This review has customer reviews, possible side effects, and important considerations about Belviq. The top 10 list featured here showcases the best diet pill supplements.
Do You Know the Best Weight Loss Pills of 2024?
Belviq Ingredients and Side Effects
| Lorcaserin Hydrochloride | Silicified Microcrystalline Cellulose | Hydroxypropryl Cellulose NF | Croscarmellose Sodium NF | Colloidal Silicon Dioxide NF |
| Polyvinyl Alcohol USP | Polyethylene Glycol NF | Titanium Dioxide USP | Talc USP | FD&C Blue #2 Aluminum Lake |
| Magnesium Stearate NF | ||||
Lorcaserin Hydrochloride: This drug is made by Arena Pharmaceuticals; it’s used to help change chemicals in the brain to suppress appetite. It can lead to addiction, and because of this it’s highly advised to make sure one follows the strict advice of a medical professional before using.
It’s also recommended to contact one’s doctor if weight loss up to 5% isn’t seen in the first 12 weeks.
Reported side effects may include:
- Extreme mood swings and unusual behavior and thoughts.
- Feeling disconnected from one’s body, suicidal thoughts, and agitation.
- Hallucination, fever, and rapid heart rate.
- Tremors, confusion, and stiff muscles.
- Swelling in the hands and feet, body ache, and flu like symptoms.
- Irritability, dizziness, and low blood sugar.
- Back pain, fatigue, and headaches.
- Dry mouth, back pain, and coughing.
There’s also other serious side effects which may result. Certain symptoms require medical attention as they can be potentially fatal. These kinds of side effects require one to immediately stop taking Lorcaserin. Follow the link provided here to find the top 10 rated diet pills.
It’s also necessary to add diet and exercise, and for some people to monitor their blood sugar. People with certain conditions may be advised to avoid using Lorcaserin altogether. A report by the FDA states that Lorcaserin was linked to:
“a number of malignant tumors”
A FDA study into this ingredient showed that it could potentially be a cancer causing drug. That’s why when this drug first appeared on the market, the FDA did not approve of it.
It’s also advised to not increase dosage of this, and to make sure to keep close contact with a medical professional in order to reduce the risk of symptoms. Certain medications and supplements may also negatively interact with Lorcaserin.
FD&C Blue #2 Aluminum Lake: Artificial food coloring that is added in tablets and capsules. It has no weight loss effect, and it’s only added to help provide a unique color.
Food colorings have been criticized due to their potential health damaging effects. The link provided here has the top 10 ranked diet pills.
EDITOR’S TIP: Combine this supplement with a proven fat burner such as Sletrokor for better results.
Belviq Quality of Ingredients
 10 of the 11 ingredients are inactive and have no weight loss purpose. The sole key active ingredient is a drug that works deep within the brain to help suppress appetite. However, it’s impossible to determine what kind of an effect this may have on health.
10 of the 11 ingredients are inactive and have no weight loss purpose. The sole key active ingredient is a drug that works deep within the brain to help suppress appetite. However, it’s impossible to determine what kind of an effect this may have on health.
It’s been shown to produce many unwanted symptoms that can be potentially life threating. It also has been shown to produce many different types of uncomfortable symptoms.
Use of this drug also requires a diet and exercise plan, as on its own it may not be effective. The DEA also considers this to be a schedule 4 drug, meaning it may be habit forming and can produce hallucinations.
 Consumer Reports has also studied this ingredient and they say:
Consumer Reports has also studied this ingredient and they say:
“People who take the drug for a year can expect to lose just 3 to 3.7 percent of their weight, and may gain the weight back, research suggests”
They also mention the possible risks:
“the drug can cause several side effect”
“The FDA’s advisory panel initially rejected Belviq because of a risk of tumors found in animal studies”
It’s because of all these reasons that they say:
“benefits are usually minimal, and their adverse effects can be troublesome”
There are many possible issues with Belviq, and there’s no guarantee of weight loss or safety. At most, the manufacturers suggest an average of 5% reduction in weight. This does not mean it will be long lasting.
The list of the top 10 diet pill supplements is available when you click here.
The Price and Quality of Belviq
Prices vary on Belviq, but a standard price listed on Drugs.com has the drug selling for $239.06 per 60 tablet bottle without a prescription. At the standard 2 pills a day, each bottle lasts 30 days and averages to a daily price of $7.96.
This is a particularly expensive diet pill, and many considerations need to be taken before using this. It’s important to follow the strict advice of a medical professional in order to avoid possible side effects. Still, there’s no guarantee of Belviq being able to produce safe and potent weight loss.
A clinical study performed by the company showed that:
“Almost half of people taking BELVIQ lost 5% or more of their body weight after 1 year of treatment”
This means that a person weighing 200 pounds would lose 10 pounds in a year, which is 1.2 pounds a month. This isn’t much when you consider the amount of effort needed and possible symptoms which may cause serious damage. Drugs.com mentions that it may cause:
“depression, suicidal thoughts or behavior, and/or any unusually changes in mood or behavior”
This amount of weight loss is also not guaranteed, as the manufactures mention to discontinue use if 5% of weight loss is not experienced within 12 weeks.
Due to it being a prescription drug, no money back guarantee is offered. This drug is particularly pricy for what can be a potentially harmful sole active ingredient. The top 10 list has the best diet pill supplements available now.
Business of Belviq
The owners of Belviq are named Eisai Co. Ltd; they can be reached via the following contact details:
Phone Number: (201) 692-1804
Address: 100 Tice Blvd
Woodcliff Lake, New Jersey 07677
This pharmaceutical company’s headquarters are based out of Japan. They have had a history of suing the FDA and DEA due to the long patent process on their drugs.
Early studies into Belviq showed that it caused tumors in rats.
Since Belviq is a prescription drug, there is no money back return policy, and no guarantee of safety.
EDITOR’S TIP: For the best results, our experts recommend using weight loss pills for at least 3 months. Save your money by buying a few bottles at once.
Customer Opinions of Belviq
Here are some reviews from online users:
“Absolutely no reaction from this drug for appetite control or energy”
“cravings shot through the roof with this medication, therefore gaining weight and excessive headaches”
“it just made me gain more weight”
“Belviq not only makes me sleepy but minor headaches, dry mouth, no constipation but rather loose stools”
Side effects were a constant issue for users. Many experienced not just physical effects but mental changes that were unpleasant. There were issues from some users who actually gained weight even while adding diet and exercise.
There were also concerns of serious side effects including mood swings, ill feelings, and many other symptoms. These kinds of issues have all been listed as possible on the official website. This is because a prescription drug has to mention all its possible side effects, in case people might have sensitivities.
Healthy customers have also experienced harsh side effects. Customer approved brands are featured via the top 10 ranked diet pills.
Conclusion - Does Belviq Work?
Before making a decision on Belviq it’s important to look over the kinds of ingredients used, what customers had to say about it, and the likelihood of weight loss effects. One has to have a certain BMI and or have an obesity related illness, while still being healthy enough to not have medical complications. Diet and exercise are required, and there’s no fat metabolism increase or fat burning effect, it’s only made to help suppress appetite. Many customers have complained that it gave them ill feelings, and that it promoted negative mental symptoms. There’s also no money back return policy offered, and this drug has been shown in some people to promote serious side effects.
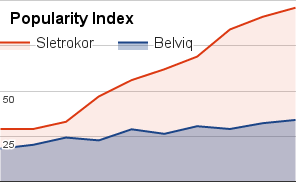
The 2024 diet pill of the year is called Sletrokor, and it’s available without a prescription. This all natural diet pill supplement has only plant based extracts, no stimulants, and it’s meant to target weight loss in many different areas.
Sletrokor is made to suppress appetite, increase metabolism, burn fat, improve mood, improve digestion, and provide many other benefits. That’s why so many customers have left great reviews. Testimonials are offered on the official website which showcases people’s results. Many have lost weight by using Sletrokor, and you can read about these results by clicking here.
Sletrokor is also offered with a full 30 day money back guarantee. If for any reason you fail to see weight loss, you can schedule a return. To learn more about Sletrokor, you can click here.
- Product Name
- User Rating
- Has Good Appetite Suppressant
- Has Only Natural Components
- Contains No Coffee
- Sletrokor
(Editor’s Choice) - 94/100
-

-

-

Belviq is new prescription drug that has been approved by the FDA under the name Lorcaserin. This weight loss drug is manufactured by Arena Pharmaceuticals, Inc. and received approval in June of 2012. Although Lorcaserin is the official drug name, it will be sold to consumers under the name Belviq. This is the first time a weight loss drug has been approved by the FDA since 1999 when Alli was approved. Belviq will require a prescription from a doctor and is designed to be used with a low-calorie diet and exercise program. Some of the symptoms that may lead a doctor to prescribe Belviq are being an adult with a BMI greater than 30. Those with a BMI above 27 along with at least one co-morbid condition like high blood pressure, diabetes or high cholesterol would also be eligible.
Lorcaserin, which will be sold as Belviq, works as a selective 2C receptor agonist. This receptor works on the areas of the brain associated with appetite control and metabolism. Stimulating the 2C receptor has been connected with reducing feelings of hunger and increasing satiety. A large scale case study was performed using Lorcaserin, in which participants lost five percent of their body weight compared to those in the study that took the placebo. The patients taking Lorcaserin also saw improvements in their cholesterol, triglycerides, HDL cholesterol and insulin resistance. All clinical trials lasted for at least one year and some participants took Belviq for two years.
Three trials were conducted, two without patients who had diabetes and one with patients with diabetes. All patients received one-on-one counseling about a reduced calorie diet and exercise at first dose of medication and every four weeks after. At one year, 47.1% of patients had lost greater than or equal to five percent of their body weight in the Belviq group and 22.4% of patients had lost greater than 10% of their body weight. While in the placebo group which is those who did not receive the study drug, 22.6 % had lost greater than or equal to 5% of their body weight and 8.7% had lost 10 or greater percent of their weight.
As part of the FDA approval, Belviq (Lorcaserin) will have to undergo six post-market studies to monitor the drug's effects on heart health.
Do You Know the Best Diet Pills of 2024?
- May help those who are obese or very overweight gain control over their health
- FDA-approved
- Prescription only
- Produces minimal side effects in comparison to other medically-prescribed weigh loss drugs
- Recommends following a healthy diet and regular exercise regimen
- Long-term health effects are still unknown after FDA-approval
- Does not address real issues of overeating
- Can be mistakenly used as a quick fix for weight loss
- Very modest weight loss in comparison to other prescription weight loss pills and diet and exercise alone
- May affect memory and attention levels
- Patients may experience withdrawal-like side effects when medication is stopped
- Lorcaserin was previously rejected due to a cancer signal being detected
Dr. Sarah G. Khan, contributing pharmacist for DietsInReview.com
- Belviq (lorcaserin) should NOT be taken in pregnancy.
- Patients should discontinue if they do not lose 5% of the body weight in the first twelve weeks of use. Duration of therapy and whether it should be weeks or 1 to 2 years has not been determined.
- Use caution with other drugs that effect serotonin levels such as antidepressants (Paxil, Zoloft, Cymbalta, Effexor etc.). There is a risk of Serotonin Syndrome, which is a rare condition that can lead to seizures. Symptoms of serotonin syndrome include increased heart rate, fluctuating blood pressure, lack of coordination, nausea, vomiting and diarrhea. If someone seems to have altered mental status they need to stop taking Belviq immediately.
- Male patients may also experience priapism, which is an erection lasting longer than 6 hours. If patient experiences an erection longer than 4 hours they should discontinue Belviq and go to the emergency room.
- Belviq increased prolactin levels which could lead to breast development in males or lactation in females. If you begin to experience these effects your prolactin levels should be monitored.
- Belviq can also have an effect on white and red blood cell count. Bloodwork may be necessary.
- If patients develop any valvular heart disease they should discontinue use. This was seen in another serotonin receptor medication that activated the 2B receptor, Belviq activates the 2C receptor.
- Belviq may cause memory and attention problems so caution should be used when operating a vehicle or machinery.
- When used along with diabetes medications, patients may experience low blood sugar. More frequent monitoring of blood sugar may be appropriate and patients should carry a small carbohydrate snack at all times to avoid this.
- It is also not advised that patients take other weight loss products along with Belviq as we do not know how they will react together.
- Safety has not been established in patients under 18 years old.
Patients taking Belviq will be advised to use the pill along with a healthy, reduced-calorie diet.
The dosage for the drug is 10 mg twice per day and can be administered with or without food. Make sure to store it in an area that is room temperature and can avoid heat or moisture (avoid medicine cabinets in the bathroom).
For best results while taking Belviq, patients should follow a regular fitness regimen.
As one of three prescription weight loss pills that have been undergoing FDA trials and review in 2010, Lorcaserin (Belviq) works to suppress the appetite by increasing the amount of the feel-good hormone serotonin, in the brain. This medication does currently boast the fewest number of side effects when compared to other prescription pills. However, the overall results are not groundbreaking.
lorcasrin, lorcserin, lorsacerin, lorsarin, lorserin, loriserin, belviq, belvick, belvic
Featured Diets and Supplements
Belviq?







 User rating
User rating


User Feedback
(Page 1 of 1, 3 total comments)Melanie
+Great drug. Very safe and effective. Highly recommended.
posted Dec 25th, 2014 11:04 amValerie
-This diet pill did absolutely nothing. If I didn't know better I'd swear it was a placebo! I lost 55 lbs on Qysmia but my body grew too used to it and I stopped losing with 15 lbs to go so I tried Belviq. Total waste of money and time!
posted Dec 4th, 2014 2:36 ampamela iribarren
como consegir el medicamento
posted Jul 17th, 2012 6:29 pm